Just out in the latest issue of FEMS Microbiology Letters is a small commentary by myself and Artist Anna Dumitriu, describing our experience of collaborating on Microbiology/Art public engagement pieces. We focus on the ethical issues involved in displaying bacteria publicly, how to address the risks involved, and projects that can open up wider debates around the ethics of new technologies, namely the Trust me I’m an Artist collaboration.
Can we display bacteria from poop? If we do, how can we minimise the risk? And who gets a say in whether we should or not?
The shiny full published version can be found: at https://academic.oup.com/femsle/article/365/11/fny101/4983122#116947251
Accepted manuscript for those who don’t have access to paywall can be downloaded here: FEMSLE_accepted_manuscript
Figures:

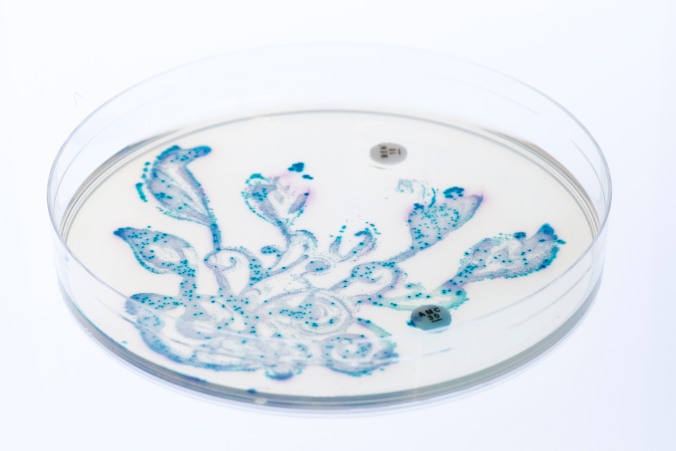
Figure 1: The MRSA Quilt The quilting squares were added to Methicillin-resistant Staphylococcus aureus cultures on chromogenic agar. Standard antibiotic susceptibility testing equipment was used to create the patterns, together with other known antimicrobial pigments and dyes. The whole work was autoclaved prior to display. It serves as a tactile, aesthetic ‘conversation piece’, as microbiological techniques are described to explain how the effects are achieved. More information on this piece and more can be found at http://www.normalflora.co.uk

Figure 2: Sequence Dress The dress was created using material impregnated and patterned using Staphylococcus aureus culture on chromogenic agar. DNA from Staphylococcus aureus cultured from Dumitriu’s body was sequenced using an Illumina Miseq, and the light output from the flow cells captured and mapped digitally onto a dress. The DNA sequence was projected behind. The piece has been exhibited internationally, and serves to highlight new technologies in the field of Microbiology. http://www.normalflora.co.uk
This is a pre-copyedited, author-produced version of an article accepted for publication in FEMS Micro Letters following peer review. The version of record [Nicola J Fawcett, Anna Dumitriu; Bacteria on display—can we, and should we? Artistically exploring the ethics of public engagement with science in microbiology] is available online at: https://academic.oup.com/femsle/article/365/11/fny101/4983122#116947251 .